Bcs Classification System
Posted : admin On 5/15/2019- Bcs Classification System
- Biopharmaceutics Classification Bcs System Class
- College Classification System
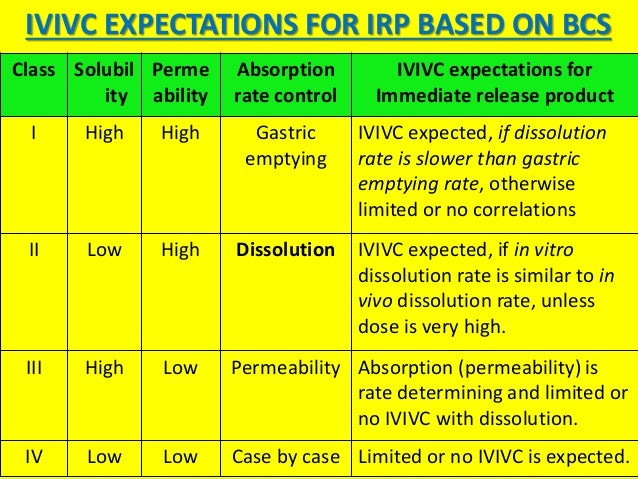
In a recent article, in vivo predictive dissolution (IPD) methodology and subclassification for the Biopharmaceutics Classification System (BCS) class II and class IV are introduced and discussed. This report proposes a simple extension of BCS class II and class IV to include subclasses of acid (a), base (b), and neutral (c). © 2011 Absorption Systems absorption.com Biopharmaceutics Classification System: Defining a Permeability Class Blair Miezeiewski, M.S. Senior Scientist, In Vitro.
- Docket Number:
- FDA-2015-D-1245
- Issued by:
The Food and Drug Administration (FDA or Agency) is announcing the availability of a guidance for industry entitled “Waiver of In Vivo Bioavailability and Bioequivalence Studies for Immediate-Release Solid Oral Dosage Forms Based on a Biopharmaceutics Classification System.” This guidance finalizes recommendations for sponsors of investigational new drug applications (INDs), and applicants who submit new drug applications (NDAs), abbreviated new drug applications (ANDAs), and supplements to these applications for immediate-release (IR) solid oral dosage forms, and who wish to request a waiver of an in vivo bioavailability (BA) and/or bioequivalence (BE) study requirement.
Submit Comments
You can submit online or written comments on any guidance at any time (see 21 CFR 10.115(g)(5))
If unable to submit comments online, please mail written comments to:
Bcs Classification System
Division of Dockets Management (HFA- 305)
Food and Drug Administration
5630 Fishers Lane, Rm. 1061
Rockville, MD 20852
Biopharmaceutics Classification Bcs System Class
College Classification System
All written comments should be identified with this document's docket number: FDA-2015-D-1245.
Regulated Product(s)